Balancing Equations Guided Practice
This is an interactive guided practice on balancing chemical equations. Good for high school and beginning college freshman level.
Plays in Web browsers on PCs and mobile devices (please use Landscape mode) so... Windows, Mac, Android phones, iPhones should all play.
It was a remake of an obsoleted old tutorial. Hope teachers and students continue to find it helpful.
Equation Categories
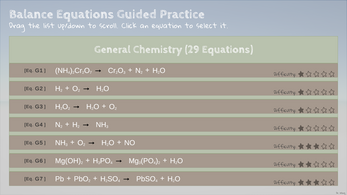
- General Chemistry
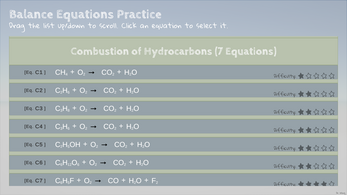
- Combustion Equations of Hydrocarbons
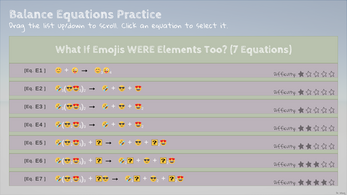
- Emoji Equations: Yep, you read it right, no kidding!
The technique used for balancing equations here is just simple math with general puzzle solving skills. It's a good exercise for your brain in general. (I think it's not even as hard as Sudoku anyway.) So why limit to real chemicals? Why not be creative and use anything fun to make balancing equations a puzzle -- a brain game? So I chose emojis...for now. 😄
How to Run the Practice:
- Select an equation from the list.
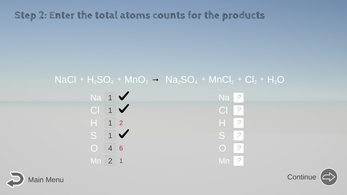
- Figure out Reactants Atom Counts: Count and enter the number of atoms on the reactant side (left side of the arrow) in the textboxes below the equation.
Click Continue to let the computer check the math for you and correct any mistakes. (If you make a mistake on an atom count, it will affect your decision to correctly balance the equation later on.) - Figure out Product Atom Count: Repeat Step 2 for the product side (right side of the arrow).
- Adjust One Coefficient: Compare the atom counts of both sides and decide on ONE atom to balance by entering ONE coefficient in the equation.
Sure, you can adjust multiple coefficients at a time and balance the equation in one shot if you want. But for beginners, try one coefficient at a time. This guided practice is designed to accommodate any skill level of fellow equation-balancers! - Update the atom counts in the textboxes as you have changed the coefficient in the equation.
- Check Your Math: Click Next/Continue to let the computer check your math for to make sure all the math of the atom counts is correct so far. (Again, if you make a mistake on an atom count, it will affect your decision to correctly balance the equation.)
- Repeat Steps 4-6 until the equation is balanced.
Some Suggestions on How to Improve Your Equation Balancing Skill
- Try practice on paper after you feel you have got the hang of it, especially if you are practicing for tests/exams that, of course, you don't get to use this guided practice.
- Note where your common mistakes are and work on those areas. For example: Is counting the atoms your biggest weakness? Was it because you often forgot to take into the account of the parentheses? Or, was deciding an coefficient for which chemical your bane?
If you always forget about the parentheses in calculating the atom counts:
- Try out the first several Emoji equation as drills.
If your common mistake is due to not knowing how to pick an coefficient for which chemical next, here are some tips:
- Choose the element that appears in only ONE chemical on each side first. It's often the metal ions, though not always.
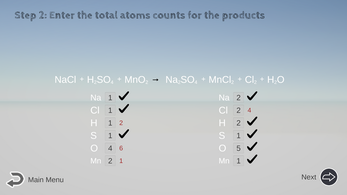
For example: NaCl + H2SO4 + MnO2 → Na2SO4 + MnCl2 + Cl2 + H2O
You would pick Na or Mn to balance first. Each of them appears in only one chemical in either side.
Next in line, the S is preferred over Cl, because S appears in only one chemical (H2SO4) in either side, but Cl appears in two chemicals (MnCl2 and Cl2) in the product side. - Aim for H and O last, especially if it has H2O.
- Try picking one atom, one coefficient at a time.
- Check your math of atom counts OFTEN: Feel free to click the Continue/Next button whenever you have adjusted anything, so you let the computer check your current math before moving on.
Why? Because... IF you make mistakes on the atom counts, you will be basing on these wrong atom counts to make a decision for the coefficients , which in this case, the coefficients very likely will be incorrect.
Examples of Choosing an Element to Balance in Some Not-So-Simple Scenarios:
Example 1: Here none of the elements is balanced.
Note that K appears in TWO chemicals in the reactant (right) side: K2Cr2O7 and KI, so it is not a good candidate to balance next.
So is Cl, which appears in TWO chemicals in the product (left) side: CrCl3 and KCl.
Therefore, for now, we eliminate K and Cl for what to balance next. You could choose from Cr, O, I, or H to balance next.

Example 2: In this scenario, Cr, O, and H have been balanced. K, I, and Cl are yet to be balanced.
Note that K appears in TWO chemicals in the reactant (right) side: K2Cr2O7 and KI, so it is not a good candidate to balance next.
So is Cl because it appears in TWO chemicals on the product (left) side: CrCl3 and KCl.
Therefore, for now, we eliminate K and Cl for what to balance next. The only good candidate to balance next is I.
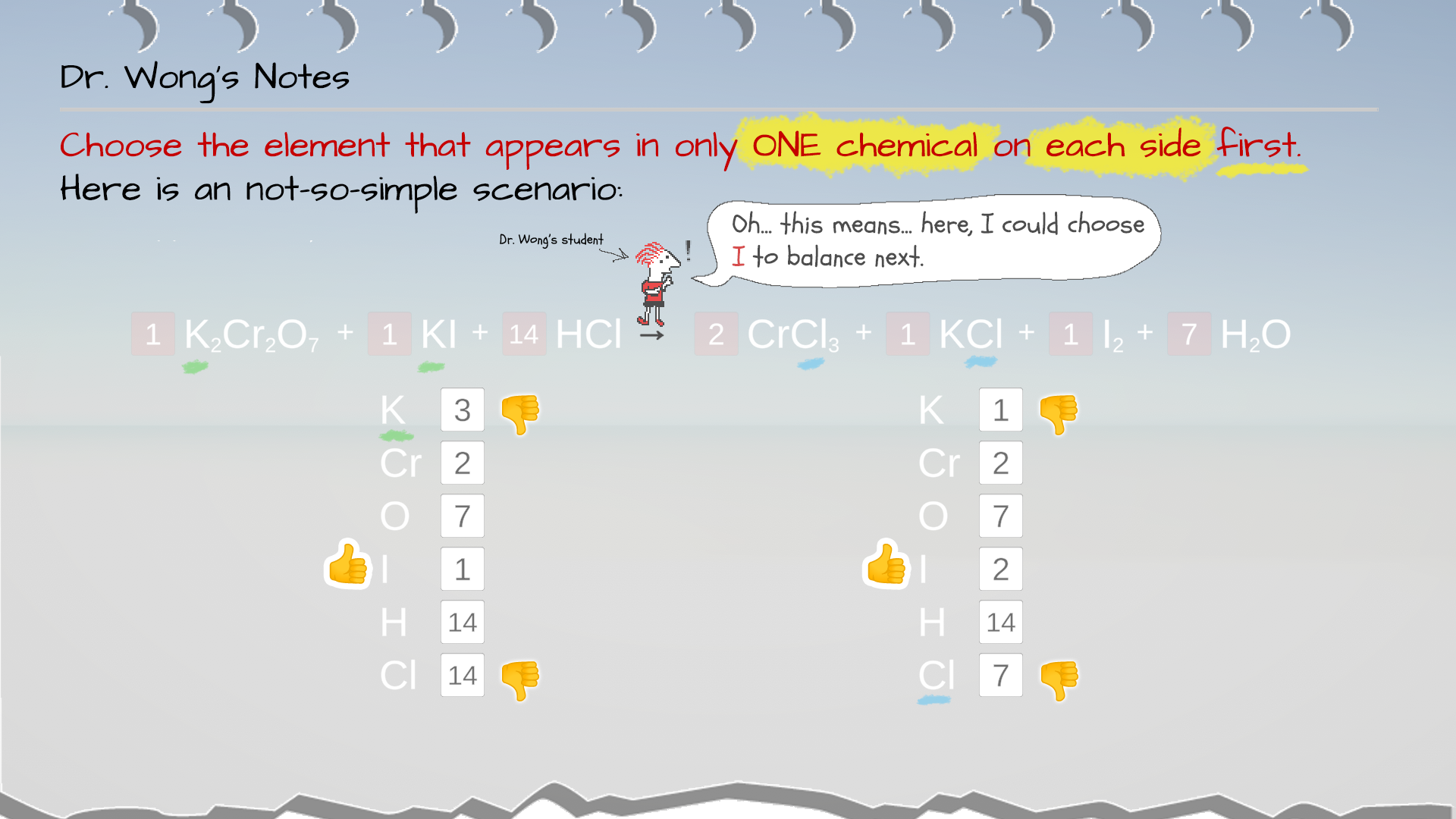
Example 3: In this scenario, Cr and O have been balanced. K, I, H, and Cl are not yet balance.
Note that K appears in TWO chemicals in the reactant (right) side: K2Cr2O7 and KI, so it is not a good candidate to balance next.
So is Cl, which appears in TWO chemicals in the product (left) side: CrCl3 and KCl.
Therefore, for now, we eliminate K and Cl for what to balance next. The good candidates to balance next is I and H.
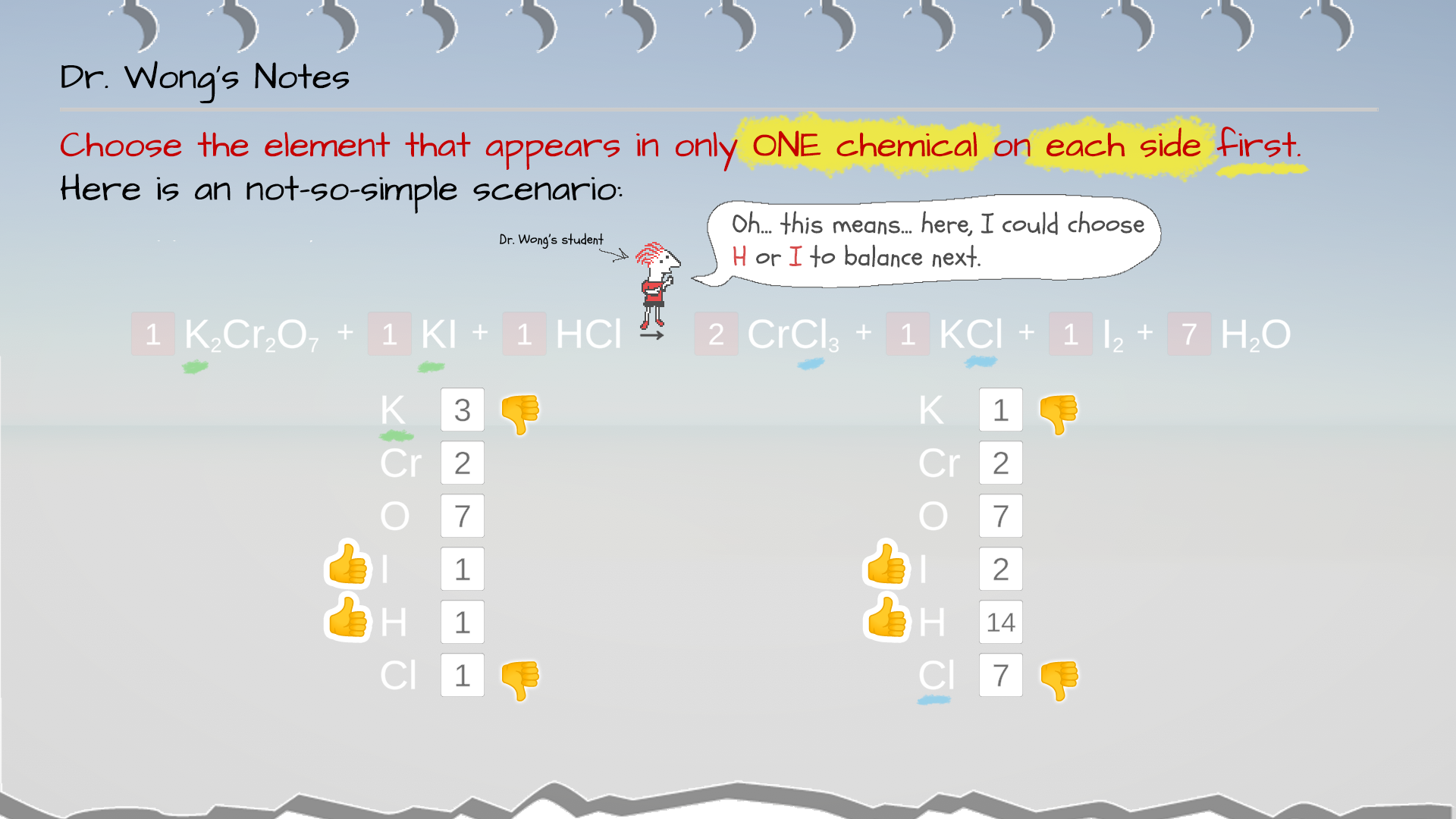
Example 4: In this scenario, Cr, O, I, and H have been balanced. K and Cl are the two remaining elements that are yet to be balanced.
But NONE of them appears only once on both the reactant and product sides.
In this case, try to pick the one that requires the simplest adjustment. The K that appears only once as KCl on the product side would be a good choice, as you could simply adjust the coefficient of KCl from 1 to 4 to make K's atom count to be 4. This will affect the Cl count on the product side, too. But this ain't the last step, and you will just have to continue the process until the equation is balanced. This is not an easy equation to balance anyway.
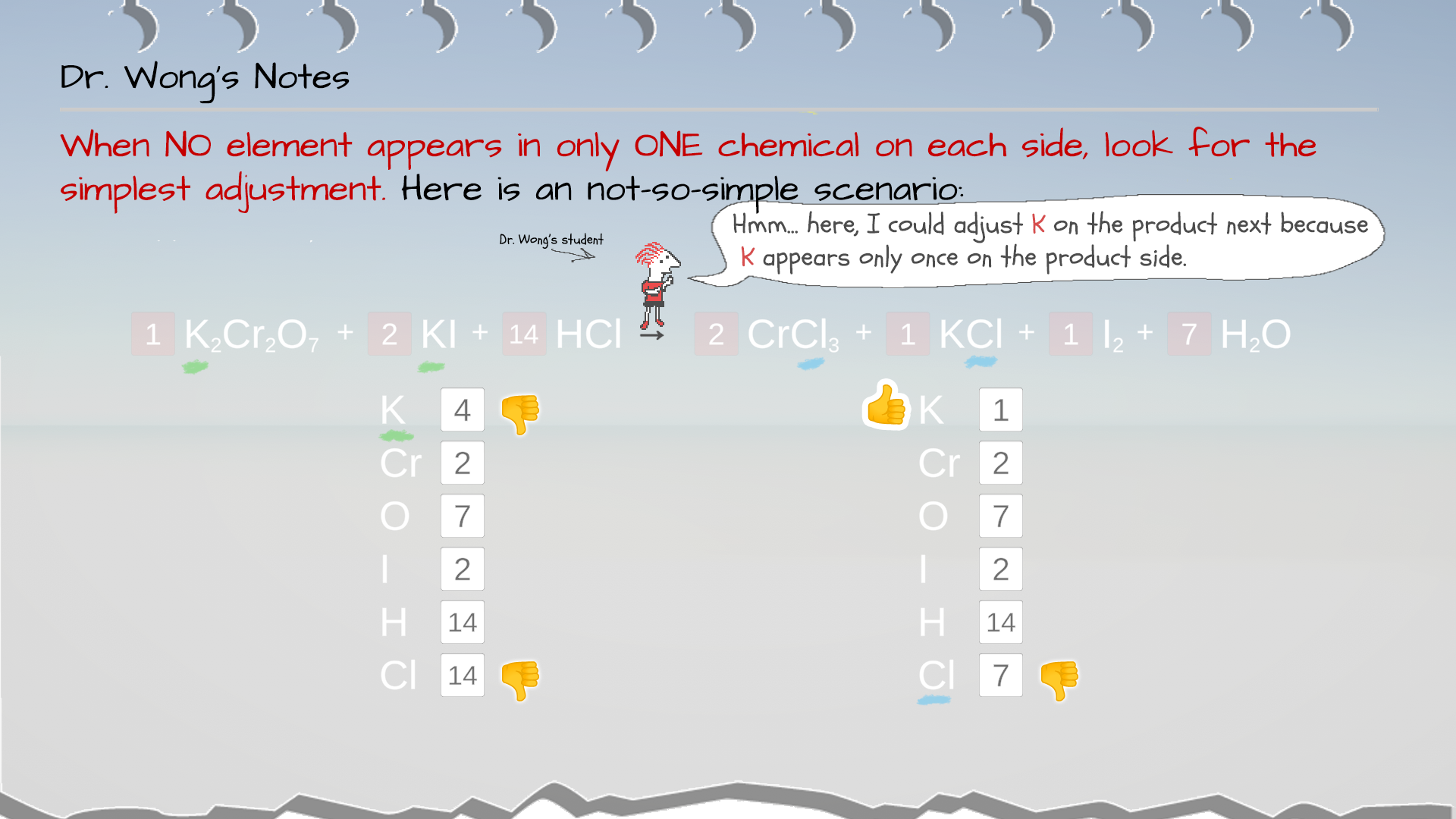
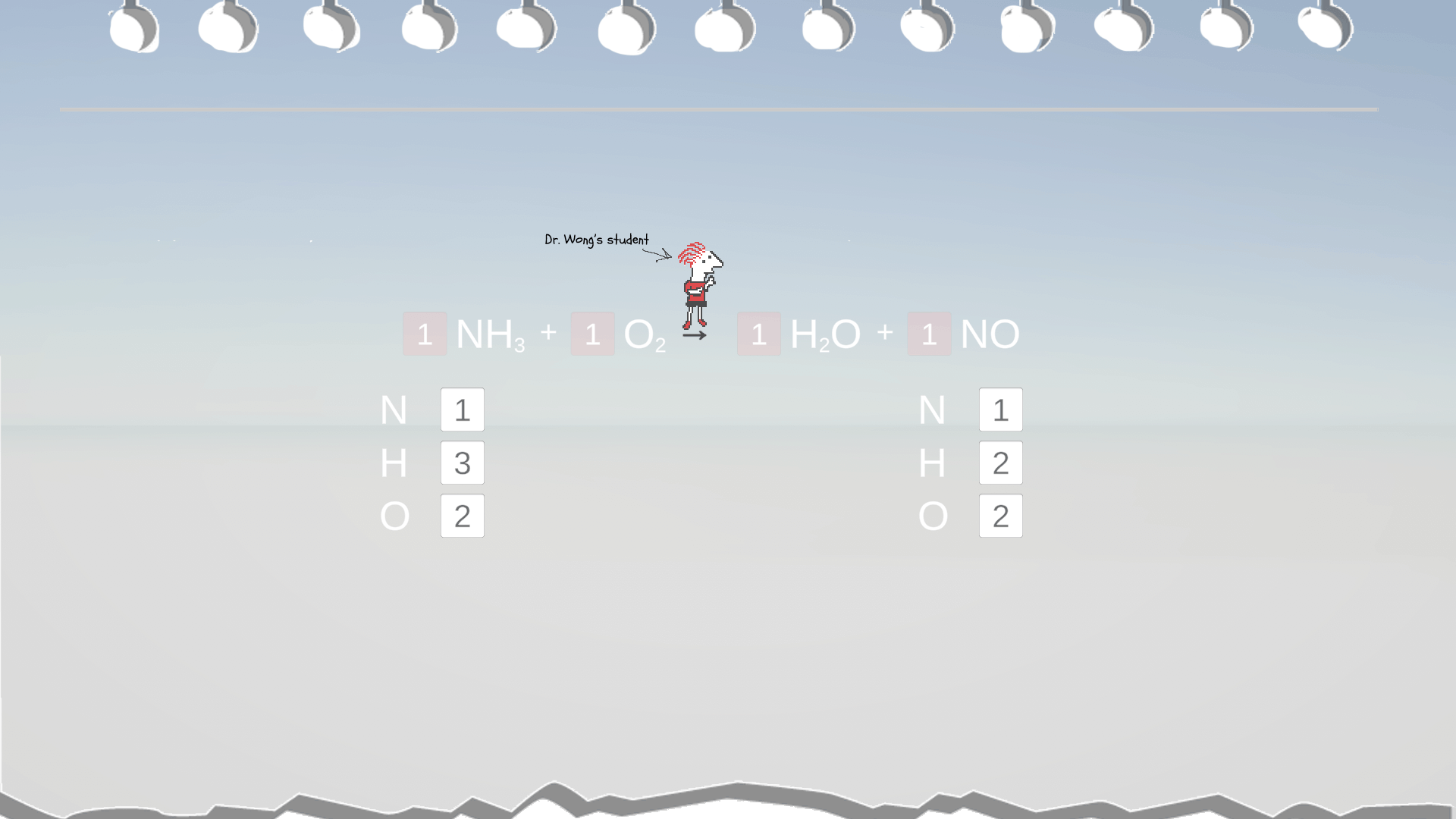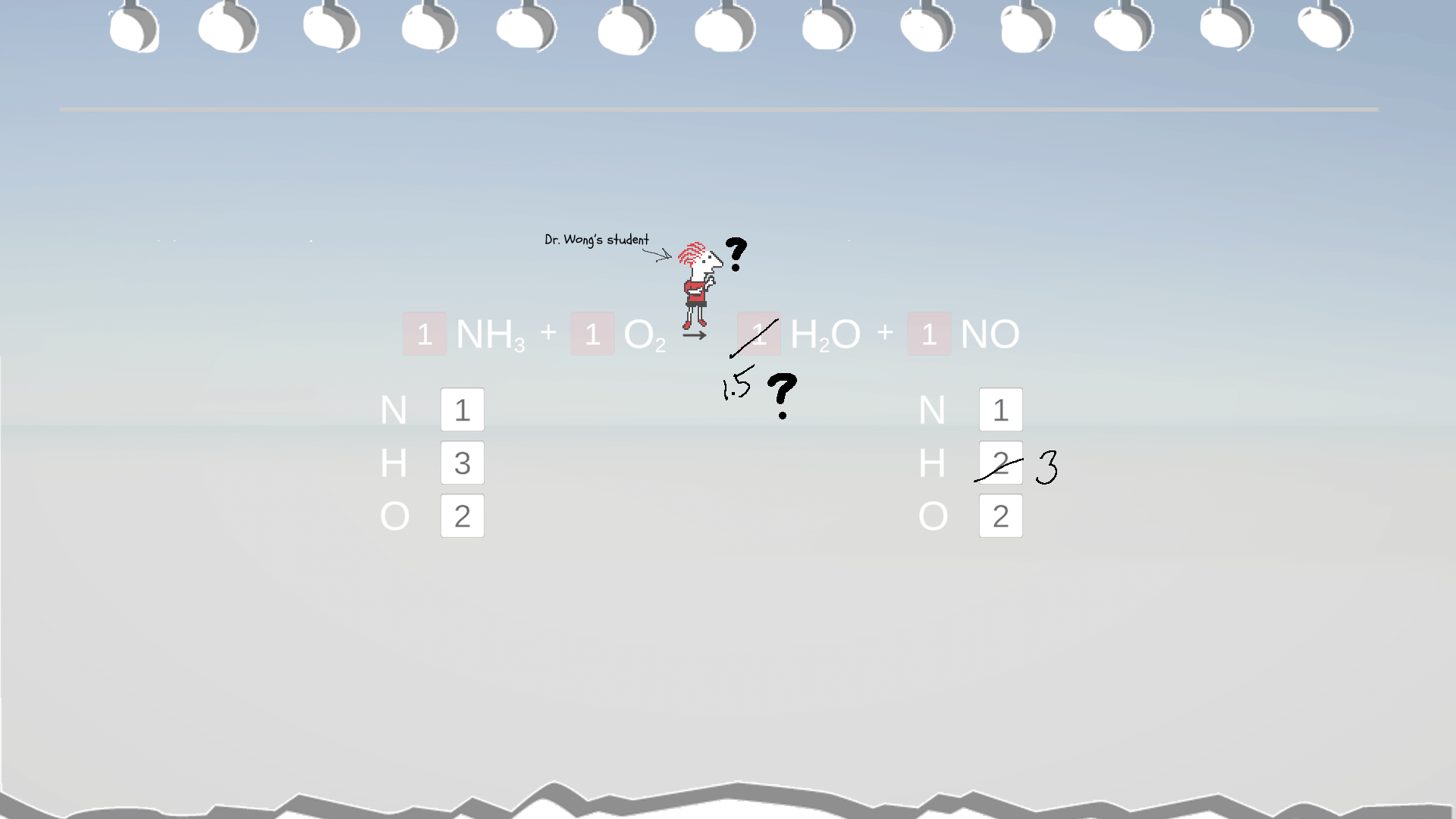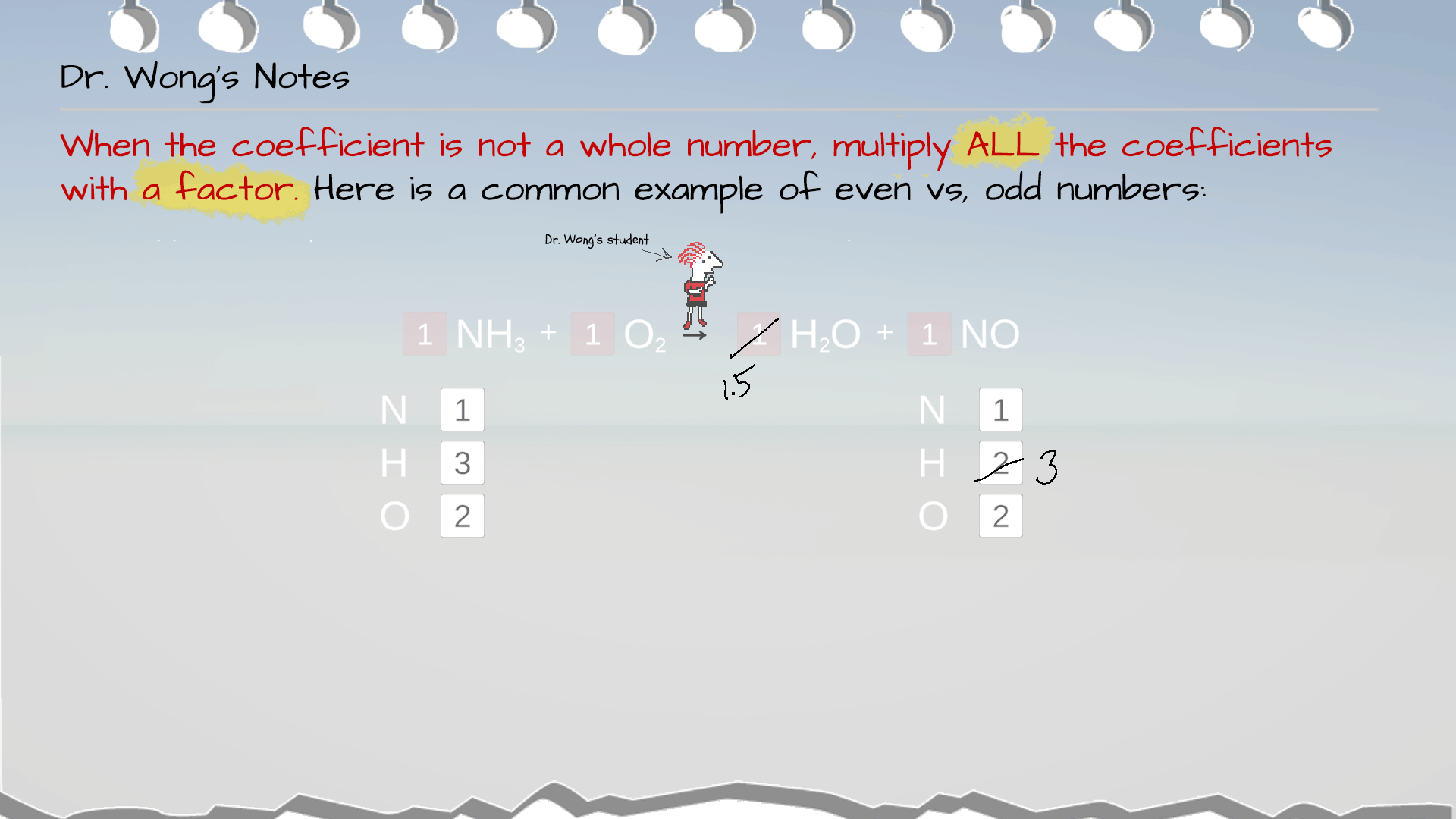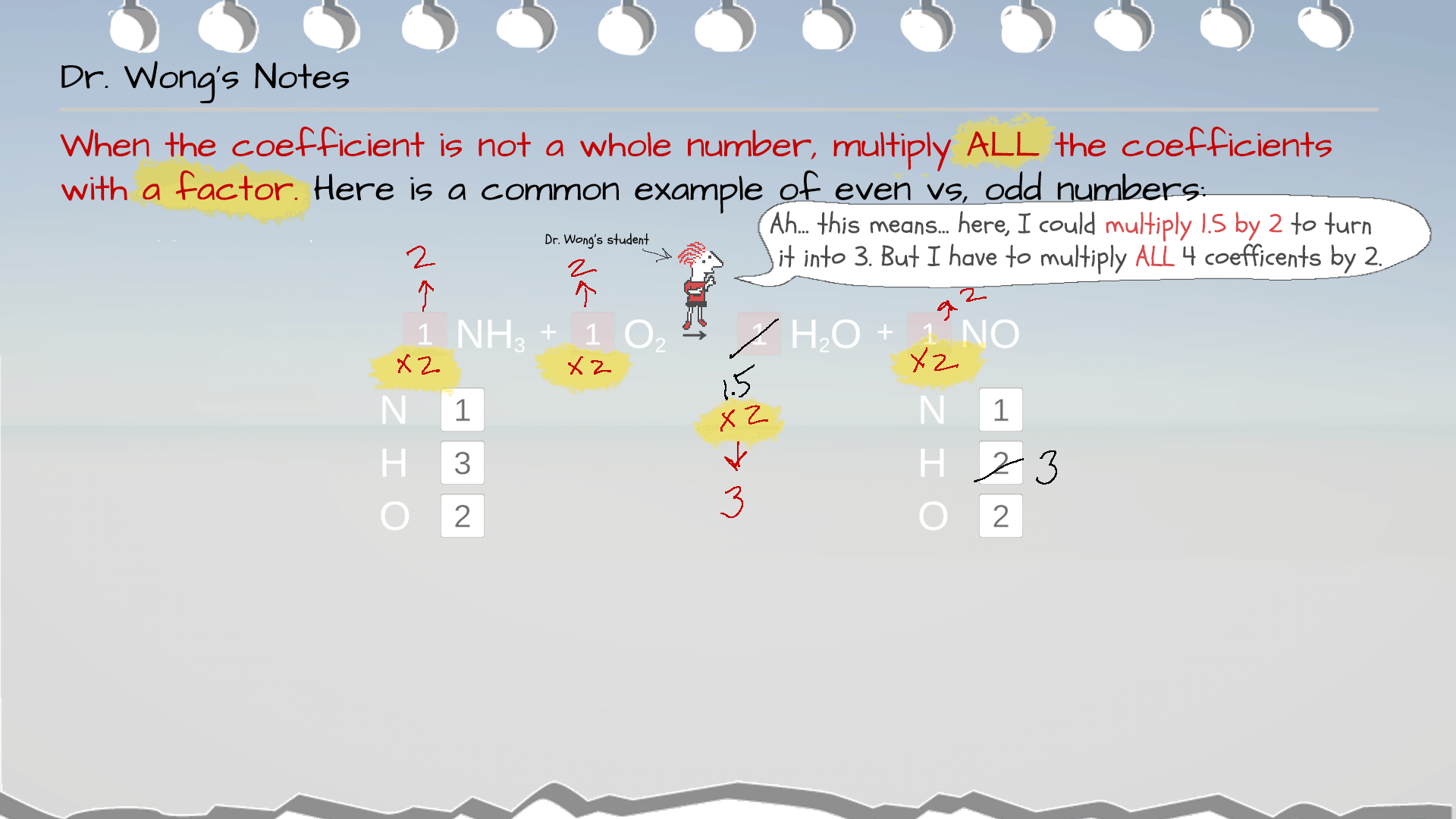
Example 5: Even Vs. Odd Atom Counts
In this example, it seems the coefficient for H2O is 1.5. However, conventionally, we use whole numbers for coefficient. So, to convert 1.5 into a whole number, the simplest way is to multiply it by 2 to make it 3. But remember to multiply ALL the coefficients in the equation by this factor of 2, too.
Additional Resource:
Check out the interactive tutorial of this balancing equation series.
| Status | Released |
| Platforms | HTML5 |
| Author | ipicio |
| Genre | Educational |
| Made with | Unity |
| Tags | balance-equation, chemistry, guided-practice |
| Average session | A few minutes |
| Languages | English |
| Inputs | Keyboard, Mouse, Touchscreen, Smartphone |